Depression is a complex, often life-altering condition that impacts how a person thinks, feels, and moves day to day. And while many treatments exist, far too many people continue to struggle from lingering symptoms, treatment resistance, or difficult side effects.
It raises an important question: are we missing a fundamental piece of the puzzle?
A growing body of research suggests we might be. Scientists are beginning to re-examine depression through a metabolic lens, looking at how metabolism, the way your body transforms energy from food and oxygen, plays a central role in shaping your mood and mental health. Within this framework, ketogenic therapy, long used for epilepsy, is emerging as a promising metabolic intervention.
In this article, we’ll explore the limitations of current depression treatments, how ketogenic therapy may address underlying causes, and what research reveals about ketogenic therapy so far.
You’ll learn:
- Why the long-held “chemical imbalance” model of depression is outdated
- Why many people don’t respond fully to conventional treatments
- How ketogenic therapy may offer a new option for treatment-resistant depression
- The essential role brain energy plays in mood and emotional stability
- The key biological pathways through which ketogenic therapy may support brain health
- How a metabolic approach to depression could unlock new possibilities for mental and physical healing
Depression: Where We Are Today
Depression affects more than 280 million people worldwide, and our current treatment landscape tells a complicated story[*]. Antidepressants and psychotherapy can be life-changing for many, but a significant number of individuals still experience only partial improvement, struggle with difficult side effects, or don’t respond to treatment at all.
This gap between available treatments and beneficial outcomes highlights the fact that our traditional models of depression don’t fully capture the condition’s biological complexity.
Today, research is revealing that depression isn’t solely a chronic chemical imbalance. It’s a condition that involves disruptions that extend well beyond neurotransmitter levels. Many patients show patterns of dysfunctional brain metabolism, including insulin resistance, reduced glucose metabolism, and chronic inflammation[*][*][*].
Taken together, these findings suggest that depression involves a fundamental dysfunction in brain mitochondrial energy metabolism, the process by which food and oxygen are converted into usable energy for the brain. This perspective opens the door to promising new therapeutic strategies.
A Word About Treatment-Resistant Depression
Within the broader landscape of depression, a substantial portion of people face an even more difficult challenge: treatment-resistant depression (TRD). Treatment-resistant depression is a term used when someone fails to respond to two or more standard treatments. Despite TRD being considered a worse form of depression, as Dr. Georgia Ede explains, TRD is actually the “norm.”
“It is more common not to respond to standard anti-depressants than it is to respond to them.” —Dr. Georgia Ede
While most people with TRD cycle through treatment after treatment, ketogenic therapy offers a unique solution because it works through different pathways than traditional approaches. By addressing brain metabolism, ketogenic therapy may be able to provide relief when standard treatments fail.
If you or someone you know struggles with treatment-resistant depression, we invite you to explore our three-part series where Dr. Bret Scher and Dr. Georgia Ede address TRD, discuss alternative treatments, and share lifestyle interventions that can significantly move the needle on depressive symptoms.
This introductory episode brings Dr. Bret Scher and Dr. Georgia Ede together to reframe treatment‑resistant depression, explain why it’s more common than many realize, and highlight the wide range of treatment options beyond standard medications.
In Part 2, Dr. Bret Scher and Dr. Georgia Ede explore a wide range of alternative treatment options for treatment-resistant depression, from neurostimulation therapies like ECT and TMS to fast-acting interventions such as ketamine, esketamine, and psychedelics, highlighting both their promise and limitations.
In the final episode, Dr. Bret Scher and Dr. Georgia Ede explore the power of lifestyle interventions, especially nutrition, sleep, exercise, and ketogenic therapy, in addressing the root causes of treatment-resistant depression and restoring brain energy.
How Could a Ketogenic Diet Help With Depression?
A ketogenic diet is any pattern of food consumption that allows your body to switch from using primarily glucose to primarily fat for fuel. In the process, the liver creates ketones, putting the body into a metabolic state known as ketosis. Ketones are an efficient, stable fuel source that the brain can use for energy, even when glucose metabolism is impaired.
By providing an alternative fuel and influencing key metabolic pathways, ketosis may help stabilize brain function in ways traditional treatments can’t. Evidence suggests that ketones could support mood regulation through several key mechanisms. Here is a breakdown of the potential mechanism for the ketogenic diet and depression.
1. Brain Energy Metabolism
A growing body of research shows that many individuals with depression experience impaired brain energy metabolism, specifically, difficulty using glucose efficiently[*][*]. This can happen in people with type 2 diabetes, metabolic syndrome, and obesity, but “brain insulin resistance” can occur even when the rest of the body is metabolically healthy. When the brain is unable to use glucose efficiently, overall energy availability to brain cells drops, and mood-regulating circuits may struggle to function at full capacity[*].
This concept aligns with Dr. Chris Palmer’s Brain Energy Theory, which posits that mental disorders stem from disruptions in the brain’s ability to produce and regulate energy. From this perspective, symptoms like low mood, lack of motivation, and emotional instability reflect an underpowered brain rather than a chemical or neurotransmitter imbalance.
Disturbances in glucose metabolism have even been linked to increased suicidal ideation and behavior, highlighting how deeply metabolic function intersects with mental health[*][*].
Ketones offer a potential solution by providing an efficient alternative fuel that bypasses insulin resistance and can stabilize the brain’s energy supply, potentially restoring function in underpowered neural circuits and supporting improved mood.
2. Anti-Inflammatory and Antioxidant Effects
Inflammation is increasingly recognized as a key player in depression, with many individuals showing elevated inflammatory markers and evidence of neuroinflammation[*][*]. In the brain, this inflammatory state can disrupt multiple systems simultaneously: it affects how key neurotransmitters are produced and used, keeps the stress-response system overactivated, and interferes with the brain’s ability to generate new cells and adapt to change. Over time, these changes can weaken emotional resilience and impair the brain’s ability to repair or recover[*].
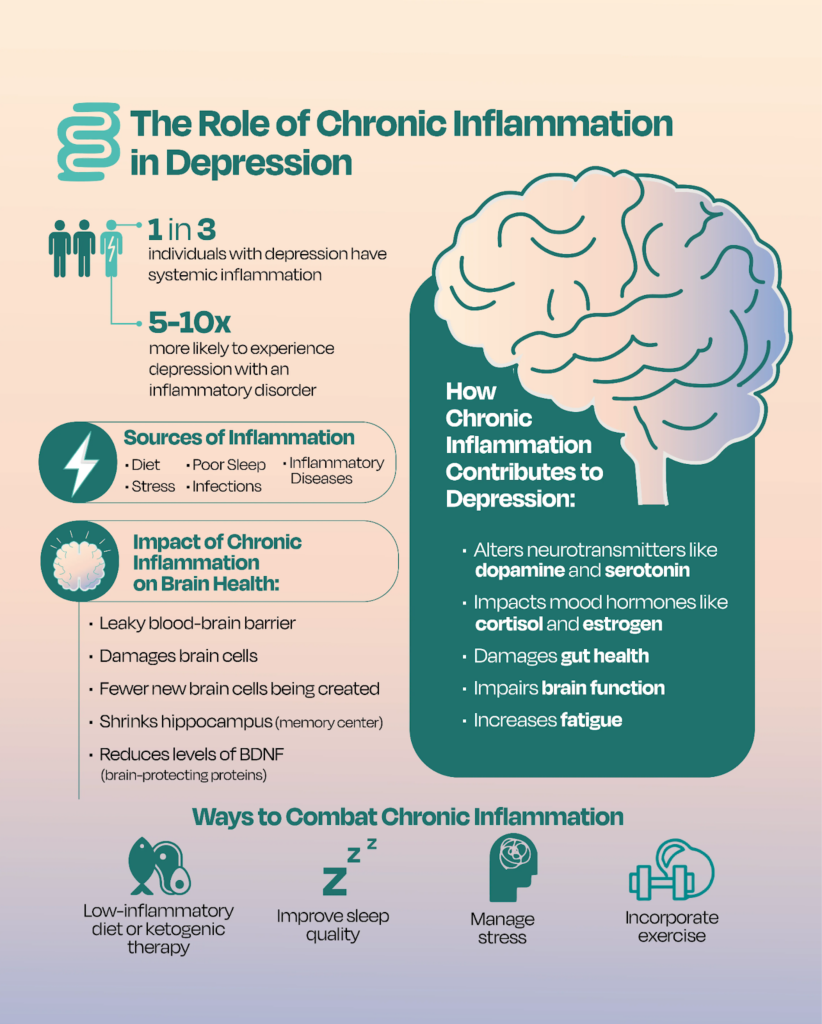
Importantly, this relationship is bidirectional. Inflammation can contribute to the onset of depressive symptoms, and depression itself can further amplify inflammatory responses due to the burden of stress on your immune system. This creates a self-perpetuating cycle that makes treatment more challenging[*][*].
Due to their anti-inflammatory and antioxidant effects, ketones may help interrupt this cycle by reducing oxidative stress and offering neuroprotection. By supporting healthier cellular environments, ketones may help stabilize mood circuits, promote resilience, and reduce the risk of ongoing neurodegeneration[*][*].
Furthermore, a well-formulated ketogenic diet is naturally rich in whole foods and low in processed foods, which often promote inflammation. In this way, the foods consumed on the diet itself offer additional anti-inflammatory benefits.
3. Increased Brain-Derived Neurotrophic Factor (BDNF)
Brain-Derived Neurotrophic Factor (BDNF) is a protein that supports brain development, plasticity, and the formation of new connections. It supports neuroplasticity (the brain’s ability to rewire itself), which is essential for learning, emotional regulation, and resilience. In depression, BDNF levels are often lower, and this reduction can make the brain less flexible and less able to recover from stress.
Many antidepressants act, in part, by increasing BDNF, which helps restore healthier brain function over time[*][*].
Emerging research shows that ketogenic therapy may have a similar effect, increasing BDNF levels and improving blood flow to the brain. In a study that examined the impact of ketogenic therapy on depression, BDNF levels rose by 32% over 12 weeks. By supporting the brain’s ability to form new pathways and repair old ones, ketones may help strengthen mood stability and overall cognitive health[*][*].
4. Autophagy Support
Autophagy is the body’s built-in cellular recycling system, an essential process that helps clear damaged components so that cells can function better. When this system becomes dysregulated, especially in brain regions connected to mood regulation, it can contribute to depressive symptoms[*].
Research in animal models shows that impaired autophagy leads to excess buildup in the brain, creating changes that are strongly linked to depression. Autophagy dysfunction also reduces the brain’s ability to regulate inflammation and may contribute to lower levels of BDNF[*].
Due to these effects, restoring healthy autophagy has become a focus of interest in metabolic psychiatry. Ketosis and intermittent fasting both appear to support autophagy, helping the brain clear cellular debris, regulate signaling, and maintain healthier emotional processing. By improving these foundational cellular processes, metabolic therapies may help address one of the lesser-known biological contributors to depression[*].
5. Gut-Brain Axis Modulation
The gut and brain are in constant conversation through what’s known as the gut–brain axis. This means that gut bacteria don’t just influence digestion; they send signals that can affect mood, stress responses, and even neurotransmitter production.
When the gut microbiome is out of balance (a state known as dysbiosis), inflammation often increases and serotonin production and signaling may be disrupted. This can affect emotional balance, as serotonin made in the gut indirectly influences the brain via the vagus nerve. And with approximately 90% of the body’s serotonin produced in the gut, shifts in the microbiome can meaningfully influence emotional well-being[*].
Interestingly, this relationship is bidirectional: psychological stress can also disrupt the gut, creating a feedback loop in which poor gut health worsens mood and low mood worsens gut health[*].
Ketogenic diets appear to shift the microbiome in beneficial ways, potentially boosting microbes that may support mood, influencing serotonin pathways, and reducing inflammation. Together, these changes may support more stable mood regulation and improved emotional resilience[*].
6. Neurotransmitter Regulation
The conventional model of depression, which focuses narrowly on neurotransmitters, is outdated, and it’s a big reason why so many people end up on multiple medications just to manage their symptoms.
That said, neurotransmitters still play a role in mood and brain function. Two of the most important are glutamate and GABA, which interact to maintain neural activity balance. Glutamate is the brain’s primary excitatory neurotransmitter, driving neuronal firing and communication, while GABA serves as its inhibitory counterpart, helping to prevent overactivation. A healthy brain depends on a stable equilibrium between the two[*].
In depression, this balance is often disrupted. Excess glutamate can create a sense of internal “overdrive,” contributing to anxiety, irritability, and difficulty regulating emotions, while low glutamate can contribute to impaired signaling within the brain. Meanwhile, too little GABA can make it harder to feel calm or grounded[*][*][*].
Another key neurotransmitter involved in depression is dopamine. Dopamine and insulin are closely linked, with insulin influencing dopamine signaling in key reward regions of the brain. When insulin signaling is impaired by insulin resistance, dopamine release may decrease, altering motivation, reward processing, and emotional resilience. All of which are features commonly seen in depression[*].
Ketogenic diets appear to influence GABA, glutamate, and dopamine in meaningful ways.
First, ketogenic diets have been shown to increase dopamine levels by modulating dopamine precursors and triggering an insulin-induced rise[*].
Ketones also naturally promote GABA production and help temper glutamate activity, reducing the risk of excitotoxicity, an overstimulation of neurons linked to mood disorders. By supporting a more stable GABA–glutamate balance, ketosis may help quiet hyperactive neural circuits and support steadier, more resilient mood states[*].
The connection between ketogenic diets and the GABA-glutamate balance is well established in epilepsy research. Dating back over 100 years now, researchers found that initiating a ketogenic state could help rebalance these two neurotransmitters in the brain and, in many cases, prevent the recurrence of seizures[*][*].
What the Research Says About Ketogenic Therapy and Depression
While ketogenic therapy may feel like a “new” idea in mental health, its roots in brain science run deep. The ketogenic diet was first introduced in 1921 as a breakthrough treatment for pediatric epilepsy, proving that changing the brain’s fuel source could profoundly impact neurological function. By 1965, researchers started exploring its potential in serious mental illness, including schizophrenia[*][*].
Research then progressed to animal models where studies demonstrated that a ketogenic diet combined with voluntary exercise reduced anxiety and depression-like behaviors in mice[*].
In the last several years, we’ve seen an increased interest in ketogenic therapy and mental health, with both clinical and case studies specifically examining the role of ketogenic therapy in the treatment of depression.
Below are a few highlights of recent research.
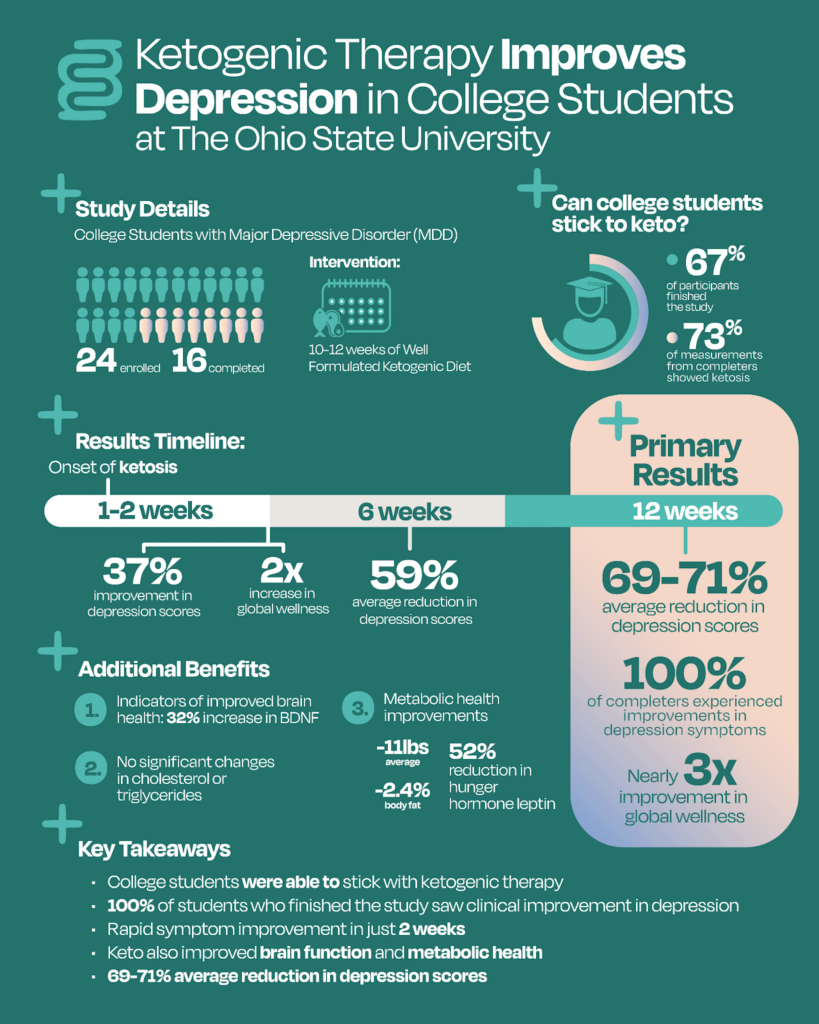
The KIND Study: Ketogenic Diet in College Students With MDD
One of the most compelling recent pilot studies on ketogenic therapy and depression explored whether a well-formulated ketogenic diet (WFKD) could serve as a feasible and effective adjunctive treatment for young adults already receiving standard care for major depressive disorder (MDD).
Sixteen students completed a 10–12-week ketogenic intervention, maintaining nutritional ketosis approximately 73% of the time. After the trial, results showed an impressive 70% drop in depressive symptoms, with improvements emerging as early as 2-6 weeks.
Beyond mood, global well-being nearly tripled, cognitive performance improved, and meaningful metabolic changes occurred. Participants lost both body mass and fat mass, and BDNF increased by 32%.
This study didn’t just change their metabolic and mental health; it truly changed their lives. As one student put it:
“Before I started the keto study, I was living every day like it was a challenge. Now, I have a reason to survive and things to look forward to, and dreams to fulfill.”
And he wasn’t alone.
“After the second week and into the third, my depression symptoms were gone. They were gone. It was incredible. I was able to jump back into my old patterns, like riding my bike to work every day and going for walks. I didn’t think I was going to be able to function like a normal person from just doing a diet. It was really mind-blowing.”
From battling depression and just trying to survive, to truly thriving, the impact that ketogenic therapy had on these young people’s lives is remarkable.
Meta-Analysis: What 50 Studies Reveal About Ketogenic Diets and Mental Health
A large 2025 systematic review and meta-analysis published in JAMA Psychiatry pulled together data from 50 studies, including randomized clinical trials, quasi-experimental designs, and case reports, to examine how ketogenic diets influence mental health.
Across different diagnoses and subject populations, ketogenic diets were linked to improvements in depressive symptoms, but one pattern stood out: the deeper and more reliably participants achieved ketosis, the stronger the antidepressant effect. Studies that used ketone monitoring and very low-carbohydrate protocols consistently showed greater improvements in mood.
Overall, the analysis reinforces a key insight that ketogenic diets may support depression most effectively when ketosis is both meaningful and measurable. This dose-response relationship underscores the importance of structured protocols, biochemical verification, and individualized support in future research.
Learn more about the JAMA Psychiatry keto and depression meta-analysis, check out our full breakdown from Dr. Bret Scher.
Case Reports and Real-World Outcomes
In addition to clinical research, there is a growing number of case reports highlighting ketogenic therapy as a successful treatment for depression and other neurological symptoms in major depressive disorder, Parkinson’s disease, and bipolar disorder.
- March 2019: The Influence of Ketogenic Diets on Mood Stability in Bipolar Disorder
- March 2022: Effects of a Ketogenic Diet on Symptoms, Biomarkers, Depression, and Anxiety in Parkinson’s Disease: A Case Study
- July 2022: Analysis of 31 Patients with Mental Illness Reports Significant Improvements in Multiple Scores of Depression
- May 2024: 3 Adults with Major Depression Experience Complete Remission of Depression and Anxiety Using Ketogenic Therapy
- August 2024: Case Study Reports Ketogenic Therapy Effectively Managed Treatment-Resistant Depressive Symptoms
Takeaway: A Promising Path Forward
Current treatments for depression often fall short of providing lasting relief. Even when symptoms improve, long-term remission is difficult to achieve, and many approaches overlook the metabolic issues that may be driving depressive symptoms in the first place.
That gap is why ketogenic therapy provides hope. It offers a low-risk, science-backed way to support the brain’s energy systems and address deeper biological factors that traditional treatments may miss.
While more research is ongoing, the current evidence makes a strong case for including ketogenic therapy as part of a comprehensive treatment strategy for depression.
To learn more, visit our Depression Topic Page, where you’ll find research summaries, expert insights, and real-world success stories from our THINK+SMART community.






